Clinical Data Management After BDS in India
Written by: Dr. Simran Jethwani
Did you know your clinical knowledge can translate into a role in pharma and clinical trials? That’s exactly where Clinical Data Management fits in.
CDM after BDS ensures that you use your clinical knowledge even beyond your clinical practice.

Introduction
Clinical data management is a systematic procedure of collecting, validating, and managing clinical trial data to ensure its accuracy, reliability, and compliance with regulatory standards.
The aim is to ensure that the data collected is of high quality and accurate, ensuring the safety and efficacy of medical trials.
To learn more about the CDM, continue reading further.
Can a Dentist Become a Clinical Data Manager in India
Yes, a dentist can switch to Clinical Data Management in India. BDS graduates can start in entry-level roles such as:
- Clinical Data Associate
- Clinical Data Coordinator
- Clinical Data Analyst.
This career path is also open to professionals from MBBS, BPT, BAMS, BHMS, Pharmacy, and Life Sciences.
With experience and skill development, professionals can move into higher roles like senior data manager and leadership positions, offering career growth and salary progression.
How BDS Skills Translate to Clinical Data Management Careers?
As a healthcare professional, clinical data management is a strong career option for you. Here are the key transferable skills you already have that can help you transition into CDM:
Transition to CDM Role
Get the roadmap
Why Choose Clinical Data Management After BDS?
Clinical data management after BDS allows you to move from treating individual patients to contributing to treatments that impact thousands globally.
CDM has an increasing scope because:
- Clinical Data Management (CDM) is a great career if you enjoy working with data, science, and healthcare.
- It plays a key role in clinical research by creating accurate and reliable data for new medicines.
- CDM professionals are involved in the entire clinical trial process, from start to finish.
- Their work helps ensure data is correct, which supports faster drug development.
- It provides strong career growth and skill development opportunities.
- Clinical data management focuses on data quality and accuracy.
- Professionals check, clean, and organise data to avoid errors in clinical trials.
- CDM is a growing and stable career field due to the rise in global clinical trials.
- Companies need skilled professionals who can follow strict data standards.
- It is a good choice if you want a career with impact, stability, and long-term growth.
Source: pmc(1)
Let’s understand what a clinical data manager does.
Is Clinical Data Management the Right Career for You?
Before switching, it’s important to evaluate if CDM fits your personality and goals.
When Should You Consider CDM as a Career Option?
You should consider CDM as a career option if you:
- Enjoy working with data and analysis
- Prefer structured, non-clinical roles
- Want better work-life balance
When Should You Not Consider Clinical Data Management?
Clinical data management might not be the best option if you:
- Enjoy direct patient interaction
- Prefer hands-on clinical work
- Dislike repetitive or data-heavy tasks
What Does a Clinical Data Manager Do?
Clinical Data Managers ensure that clinical trial data is accurate, reliable, and compliant with regulations.
The following are the roles and responsibilities of a clinical data manager:
Design and manage clinical study databases while standardising medical terminology to ensure accurate, structured, and analysis-ready data from the start.
Key Responsibilities:
- Clinical database design, setup, and maintenance
- CRF/eCRF development and validation
- Version control and database updates
Gather data from multiple sources and ensure every entry is accurate, complete, and free of discrepancies.
Roles of a CDM:
- Data entry review and validation checks
- Query generation and resolution
- Data discrepancy management
Maintain data accuracy while meeting regulatory standards and keeping all documentation audit-ready.
Role includes:
- Adherence to GCP and regulatory guidelines
- Audit readiness and documentation management
- Data quality assurance processes
Clinical data managers work closely with cross-functional teams like pharmaceutical, CRO, and biotech and provide training to ensure smooth data handling processes.
Key Responsibilities:
- Coordination with clinical research teams and sponsors
- Training site staff on data entry systems (EDC)
- Supporting study teams during trial execution
They generate reports and ensure proper database closure at the end of clinical studies.
Key Responsibilities:
- Data extraction and reporting for analysis
- Database lock and study closure activities
- Final data validation and archival
Source: linkedin(2)
A clinical data manager ensures that clinical trials are factually accurate and comply with regulatory compliance (e.g., GDPR, HIPAA) across health information systems.
Work is usually office-based or remote, with structured timelines and deadlines.

To perform these responsibilities effectively, let’s explore the essential skills required for Clinical Data Management.
What are the Skills Required to Transition from Dentistry to CDM?
Core Technical Skills for Clinical Data Managers
- Electronic Data Capture (EDC) Systems: Medidata Rave, Oracle Clinical, Inform
- Data Cleaning & Query Resolution: Detecting missing data, inconsistencies, and correcting them
- Clinical Coding Tools: MedDRA for events, WHO Drug for medications
- Excel Skills: Pivot tables, lookups, and conditional formatting
- Knowledge of CDISC standards and data interchange formats such as SDTM (Study Data Tabulation Model) and ADaM (Analysis Data Model).
Soft Skills Required to Become a Clinical Data Manager
As a healthcare professional, you already possess many of the skills required to succeed in Clinical Data Management.
Let’s take a closer look at the transferable skills that make this career transition easier.
- Attention to detail
- Analytical thinking
- Communication skills
- Time management
With your healthcare background, your transferable skills are enough to get started in Clinical Data Management.
In most entry-level roles, technical skills and tools are taught during on-the-job training, making it easier to transition without heavy upfront investment.
How to Get Your First CDM Job After BDS
Here is a step-by-step guide to landing a job in CDM after dentistry
- Build Relevant Skills
- Learn Microsoft Excel and clinical trial basics through free courses.
- Create a Strong Resume
- Highlight clinical knowledge
- Add relevant certifications
- Showcase transferable skills
- Optimise your LinkedIn Profile
- Optimise your LinkedIn profile with a targeted headline like “Aspiring Clinical Data Management Professional” to attract recruiters.
- Update the “about” section
- Network Strategically
- Connect with CDM professionals and recruiters on LinkedIn
- Actively engage with relevant content by commenting on posts and participating in discussions to improve your visibility.
- Start with Entry-Level Roles
- Clinical Data Associate
- Data Coordinator
- Junior Data Analyst
Note: Odigio Careers does not recommend pursuing paid courses unless specifically required.
Educational Pathways for CDM
The essential educational qualifications required for clinical data management can vary depending on the organisation and the job role requirements.
However, most positions require a combination of healthcare and life sciences education, as well as additional educational qualifications in computer science or domain-specific training.
An undergraduate degree is mandatory to pursue a career in Clinical Data Management.
- Healthcare– BDS, MBBS, BAMS, etc.
- Life Sciences– BPharma, BSc (biology, biotechnology, biochemistry, microbiology, etc.)
If you want to transition your career from BDS to CDM, you may lack the technical knowledge of clinical trials, data standards, or CDM-related workflow.
To gain knowledge and fill these gaps, transitioning into CDM requires.
- Industry-related knowledge
- Increased career opportunities
- Skill development in CDM tools
- Confidence in handling complex data
- Understanding regulatory compliance and best practices
*Odigio does not recommend pursuing paid courses until a specific requirement is specified.
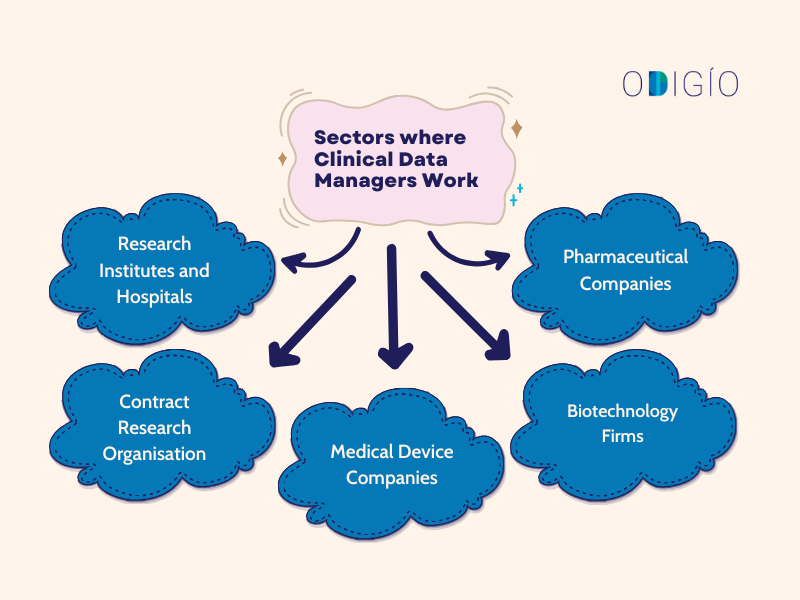
Sectors where Clinical Data Managers Work
Clinical data managers typically work in the following domains:
- Pharmaceutical Companies: Management of data from drug development. Key employers (e.g., Pfizer, Novartis).
- Biotechnology Firms– Management of data from clinical trials.
- Medical Device Companies: Analyse data related to the safety and efficacy of medical devices.
- Contract Research Organisation: Conducts clinical trials on behalf of other companies. Key employers (e.g., IQVIA, Parexel, ICON).
- Research Institutes and Hospitals: Work on different research studies conducted by faculty.
Career Opportunities in CDM
Different job roles in Clinical Data Management are as follows:
- Clinical Data Coordinator/Assistant: Handles data entry, basic checks, and query resolution.
- Data Entry Operator: Ensure accurate data entry
- Junior Data Analyst: Assists in identifying data discrepancies and cleaning data.
- Clinical Data Analyst: Cleans, validates, and analyzes trial data.
- Database Programmer: Designing databases, coding edit checks, implementing CDISC standards (using tools like SAS).
- Clinical Data Manager: Overseeing study databases, query management, and ensuring protocol compliance.
- Medical Coder: Assigning standardized codes (MedDRA, WHO-DD).
- Lead Data Manager: Supervising teams, study timelines, and database quality.
- CDM Project Manager: Managing end-to-end data processes, stakeholder communication.
- Quality Control Specialist: Ensuring regulatory compliance, auditing databases.
- Associate Director/Director of CDM: Strategic leadership, department management.
It depends on various factors, such as the company you work for, location, and your skill set, among others. (3)
Future Scope of Clinical Data Management
With advancements in AI and automation, CDM is evolving rapidly.
- AI improves data accuracy and efficiency
- Cloud-based systems enable real-time collaboration
- Demand for CDM professionals continues to grow
Take the First Step
Start here
Required Clinical Data Management Systems
Clinical Data Management (CDM) depends on management systems to ensure efficient, accurate, and compliant handling of clinical trial data.
Below are some commonly used systems in CDM.
Software and Systems Required for Data Management
The most widely used tools in CDM are as follows.
- Oracle Clinical Data Management- A robust tool offering CDM functionality, including EDC and database management.
- Medidata Rave- A leading EDC tool for designing, capturing, and managing clinical data trials.
- Clinion EDC- Simplified database capture and management for small to medium trials.
- Veeva Vault EDC- A modern cloud-based solution for managing faster clinical trials.
Technologies in Clinical Data Management
Here are some essential technologies in clinical data management that you should start getting familiar with.
- Artificial Intelligence (AI)
- Machine Learning (ML)
- Cloud-based solutions in Clinical Data Management (CDM)
Advances in technology are improving how clinical trial data is captured, managed, analysed, and reported.
These developments enhance efficiency, reduce costs, improve accuracy, and enable real-time collaboration.
Challenges in the CDM Role
Every profession has its challenges, and clinical data management is no exception. Let’s read about the challenges faced by Clinical Data Managers.
- Data Quality: Clinical trials require error-free, well-organised documentation. As a healthcare professional, you maintain patient records and follow clinical protocols, so attention to detail is a key strength.
- Regulatory Compliance: CDMs operate under the ICH-GCP, FDA, DCGI, and GDPR, among other regulations. BDS graduates already possess a solid understanding of ethics, consent, and medical regulations.
- Data Privacy: Handling sensitive patient data requires discipline and adherence to strict protocols. Medical training builds that confidentiality mindset.
- Costs and Delays in Trials: Even one incorrect datapoint can push timelines. Dentists already understand how diagnostic delays impact patient outcomes.
Now that you have acquired the basic knowledge about clinical data management, let’s explore how you can establish a career in this profession.
Continuous Learning and Skill Development
Clinical Data Management is a dynamic field that requires professionals to stay updated with evolving regulations, technologies, and industry trends.
Here are some key strategies to help you continue learning.
- Improve technical skills
- Pursue relevant certificate courses
- Improve project management skills
- Stay updated with industry standards
- Basic understanding of CDM principles
- Participate in industry training programs
Conclusion
Clinical Data Management after BDS opens doors to a dynamic career in clinical research.
Embrace learning, gain certification, and stay updated; your analytical skills can lead to impactful contributions to the healthcare field.
With advancements in AI, automation, and regulatory standards, CDM professionals ensure data accuracy and smooth functioning.
Cross-functional collaboration, combined with skill enhancement, can propel you into leadership roles, making CDM a promising career path.
Switch to CDM
Take the first step
Clinical Data Management After BDS: FAQs
Refrences
- Grand View Research: India Clinical Trials Market Size & Outlook, 2025-2033
- LinkedIn: Hiring clinical data managers
- Glassdoor: Clinical Data Managers Salaries
